Thus, the RPA-binding sites within the BTR complex have not yet been precisely mapped and the physiological relevance of its association with RPA has not yet been defined. However, later work suggested that RMI1 rather than BLM mediates RPA association with the BTR complex 19. All three subunits contain oligonucleotide (OB)-binding folds that either bind to ssDNA or mediate protein–protein interactions, with the N-terminal OB-fold of RPA1 and the C-terminal winged-helix domain of RPA2 being the major interaction sites recognized by proteins that respond to DNA damage or replication stress.īLM complexes isolated from cell extracts contain RPA 16, and in early studies the N-terminal 447 residues of BLM were suggested to bind directly to the RPA1 subunit 17, 18. It consists of three subunits, RPA1, RPA2, and RPA3 (also referred to as RPA70, RPA32 and RPA14 respectively, based on how they migrate during sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE)). RPA is an abundant, high-affinity ssDNA-binding complex that is thought to recognize ssDNA rapidly in cells whether it is generated during DNA replication or as a result of DNA damage 15.
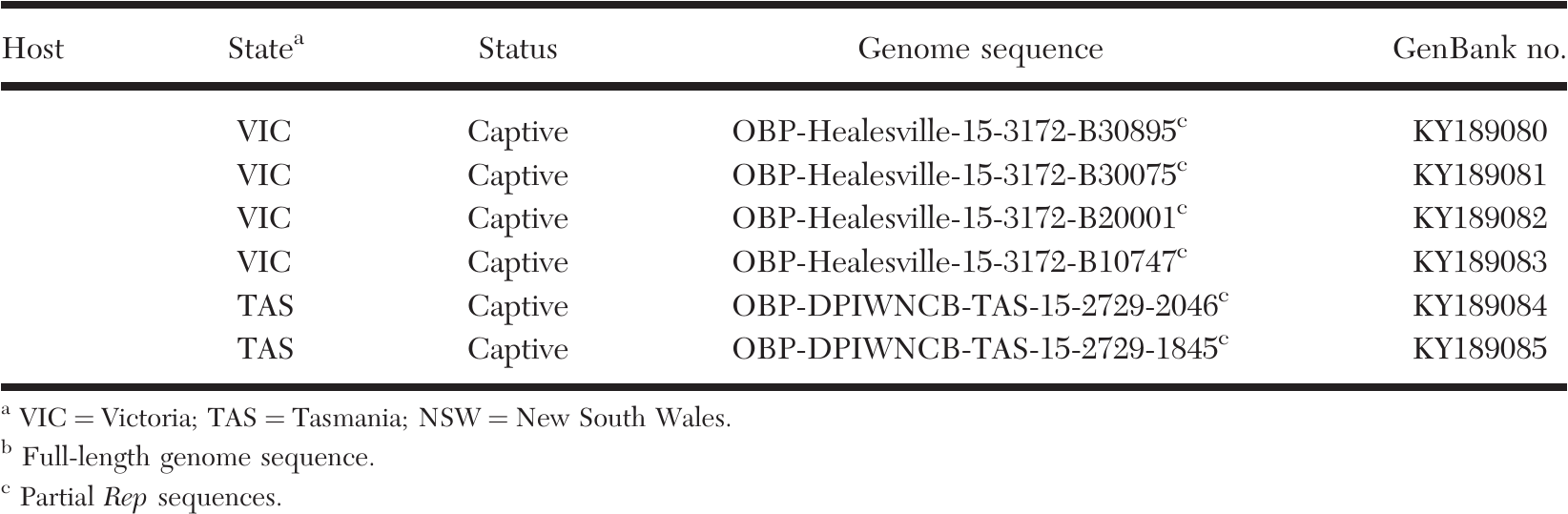
Furthermore, very little is understood as to how the activities of the BTR complex are regulated in response to genotoxic stress, and how BLM can tell the difference between normal DNA metabolic processes and DNA damage or stalled replication forks. Currently, no separation-of-function mutations have been identified that could distinguish the roles of BLM in double Holliday junction (dHJ) dissolution, DNA-end resection, UFB processing, and stalled fork restart, to prove which contribute to tumor suppression in a suitable animal model. Such functions include an early-stage role in the HR repair process by stimulating DNA-end resection 12, processing of ultra-fine DNA bridges (UFBs) between sister chromatids in anaphase 13, and promoting restart of stalled replication forks 14.

However, more recent data from genome-wide analyses suggest that loss-of-heterozygosity events in BLM-deficient cells are extremely rare 11, indicating that other cellular functions of BLM may contribute to the increased cancer predisposition in Bloom syndrome patients. BLM has been proposed to act as a tumor suppressor by preventing crossovers between homologous chromosomes, which could lead to loss-of-heterozygosity 9, 10. Accordingly, mutations in TOP3A, RMI1, and RMI2 have been identified in patients with conditions similar to Bloom syndrome 7, 8. Cells from these patients display multiple signatures of genome instability, in particular a large increase in sister chromatid exchanges (SCEs) 4, which consequently serves as a diagnostic test for Bloom syndrome 5.īLM functions in cells with TOP3A, RMI1, and RMI2, which together form the BTR “dissolvasome” complex that can process HR intermediates to prevent genetic crossovers 6.
#Victoria 2 ultimate rebooted 2018 full#
Remarkably, Bloom syndrome patients are predisposed to the full spectrum of malignancies found in the general population, in contrast to other inherited cancer-predisposing disorders 3.

It is characterized by growth retardation, immunodeficiency, hypersensitivity to sunlight, and high cancer predisposition 2. Bloom syndrome is a rare hereditary chromosomal instability disorder caused by mutations in the gene encoding the BLM helicase 1.
